What Is LuminLogic?
Last updated: May 8, 2026
LuminLogic is the premier AI-enhanced eQMS and regulatory compliance platform for MedTech. Purpose-built for medical device companies and regulated industries, it provides a centralized, auditable environment for managing the documents, processes, and records that regulatory frameworks demand — without the overhead of paper-based systems or disconnected spreadsheets.
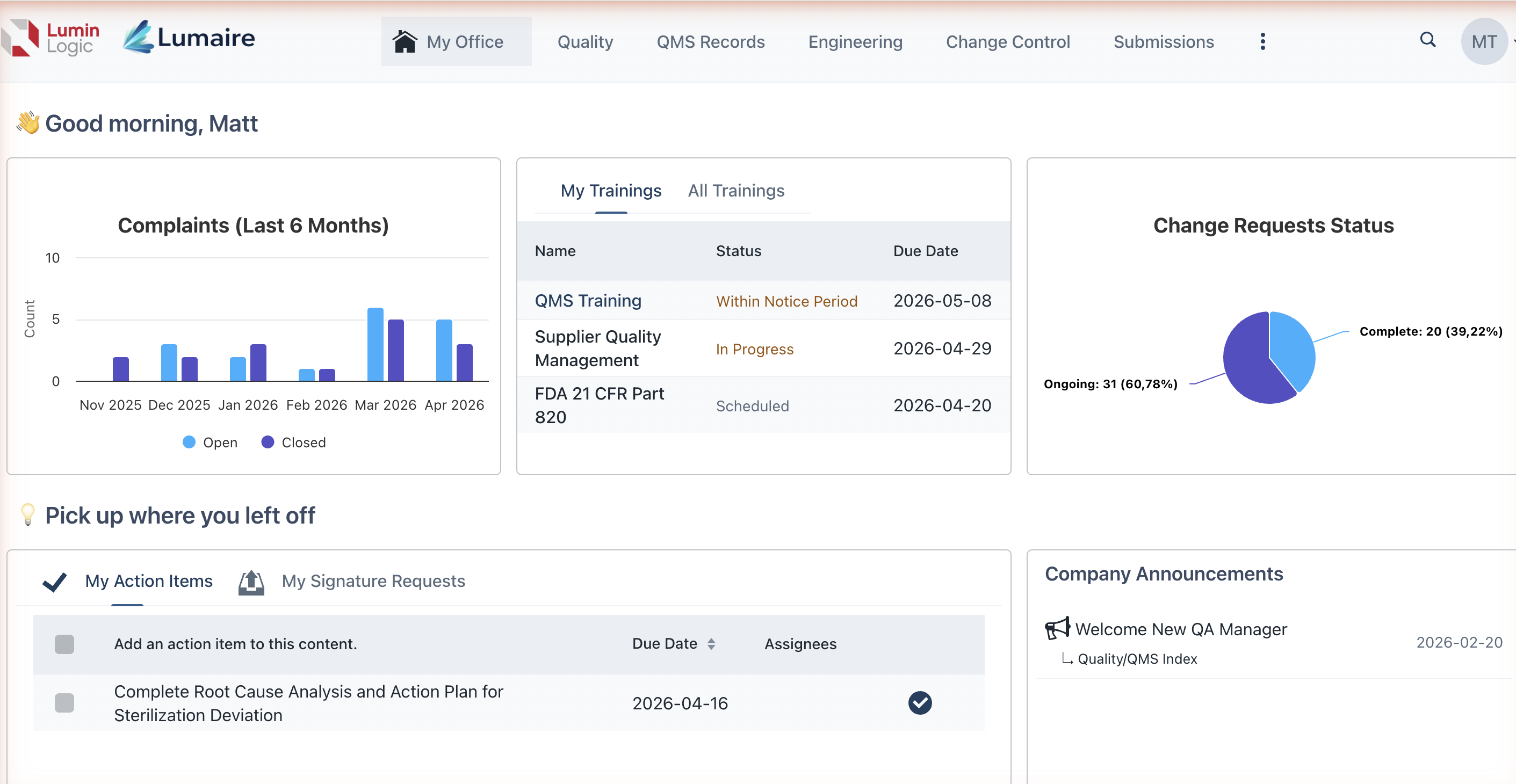
Key Capabilities
LuminLogic brings together the core pillars of a modern QMS in a single platform:
Document Management — Author, review, approve, and control documents with full version history and electronic signatures that meet the compliance expectations your auditors look for.
CAPA Tracking — Manage Corrective and Preventive Actions from identification through effectiveness checks, with structured workflows and audit trails.
Change Control — Route change requests through configurable approval workflows so every modification is evaluated, documented, and traceable.
Complaint Handling — Capture, investigate, and resolve product complaints with links to related CAPAs, risk assessments, and regulatory reports.
Training Management — Assign training content, track completion, and verify competency — critical in regulated industries where training records must demonstrate that staff are qualified for their roles.
Traceability Matrices — Map relationships between requirements, design inputs, verification activities, and risk controls to demonstrate the design traceability your auditors expect.
Project Management — Plan and track quality projects, milestones, and deliverables alongside your QMS records.
Electronic Signatures — Apply legally binding electronic signatures with full compliance support for FDA 21 CFR Part 11 and EU Annex 11, including meaning-of-signature declarations.
AI-Powered Assistance (LuminLogic IQ) — Use built-in AI tools to draft content, analyze project status, summarize documents, and accelerate routine quality tasks.
Designed for Compliance
LuminLogic is architected to support compliance with:
FDA 21 CFR Part 820 — Quality System Regulation for medical devices
ISO 13485:2016 — Medical devices quality management systems
ISO 9001 — General quality management systems
FDA 21 CFR Part 11 / EU Annex 11 — Electronic records and electronic signatures
Audit trails, access controls, electronic signatures, and controlled document workflows are built into every area of the platform — not bolted on as afterthoughts.
Blueprints: Your Starting Point
When you set up a LuminLogic workspace, you choose a Blueprint — a pre-configured template that determines the structure and content types available to your organization:
Standard — A minimal starting point for organizations that want to build their own structure from scratch.
QMS — A comprehensive quality management configuration with pre-built functional areas, document types, and workflow templates aligned to ISO 13485 and FDA 21 CFR Part 820.
Project Management — Focused on project tracking and deliverable management, suitable for teams that need structured project oversight alongside quality records.
Blueprints can be customized after setup. They are a starting point, not a constraint. See Setting Up a New Workspace for details on choosing a Blueprint during workspace creation.
Functional Areas: The Organizational Backbone
LuminLogic organizes content into Functional Areas — logical groupings that mirror the departments or process domains in your organization (e.g., Design Controls, Document Control, Supplier Management, Risk Management). Each Functional Area can contain its own set of documents, records, workflows, and access permissions. Functional Areas appear as navigation links in the top bar, giving every user quick access to the areas relevant to their role.

Multi-Language Support
LuminLogic supports five languages out of the box: English, German, Greek, Spanish, and Portuguese. Users can select their preferred language in their profile settings, and the entire interface — labels, menus, notifications — will display in that language.
Who Uses LuminLogic?
Role | How They Use It |
Quality Managers | Manage CAPAs, oversee document control, run audits, and monitor QMS health |
Regulatory Affairs | Track compliance activities, manage change control, and prepare submission documentation |
Executive Leadership | Review dashboards, approve high-level documents, and monitor organizational quality metrics |
General Employees | Complete training assignments, acknowledge documents, submit complaints, and participate in workflows |
Ready to get started? Learn how to log in to LuminLogic or explore navigating the interface.